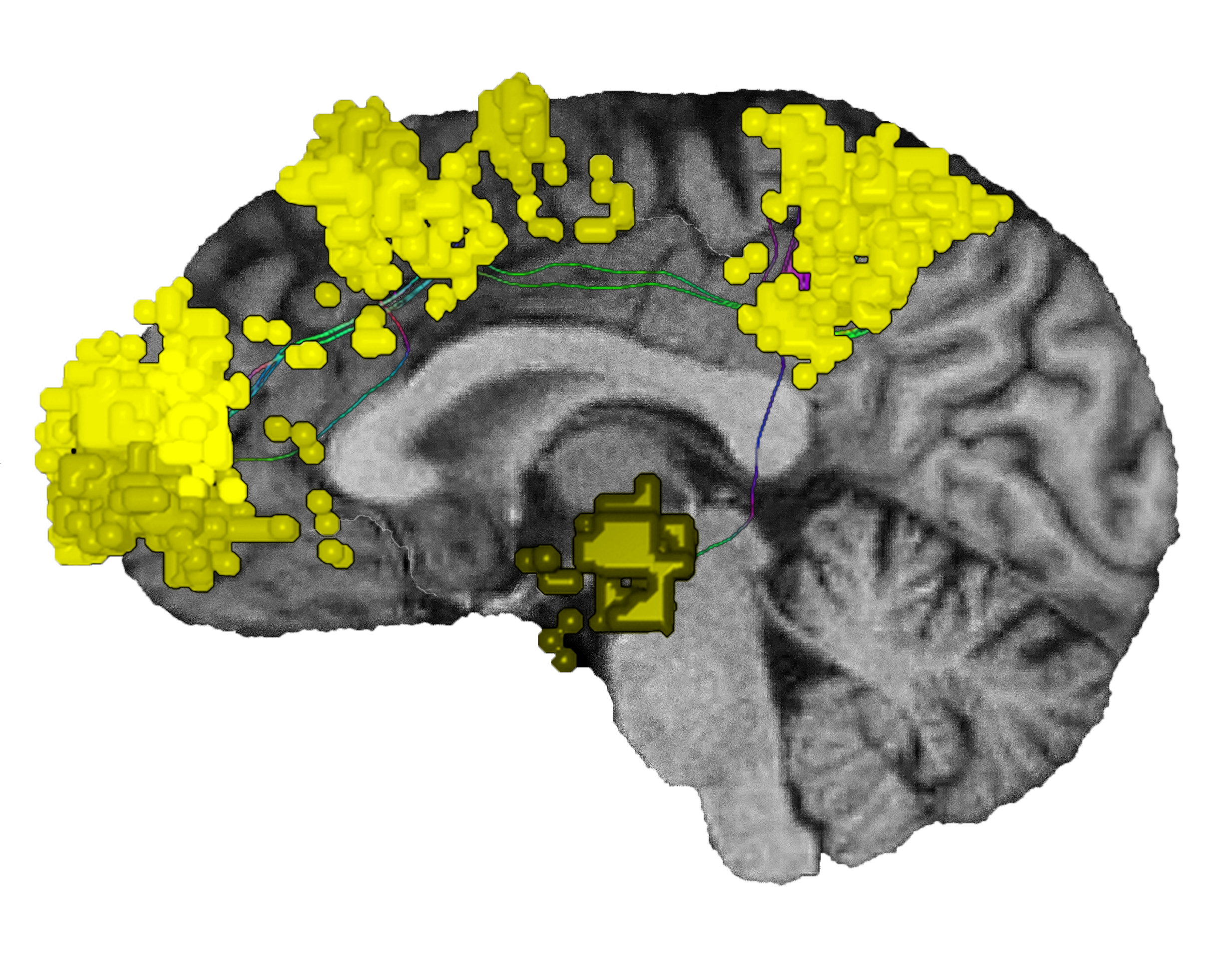
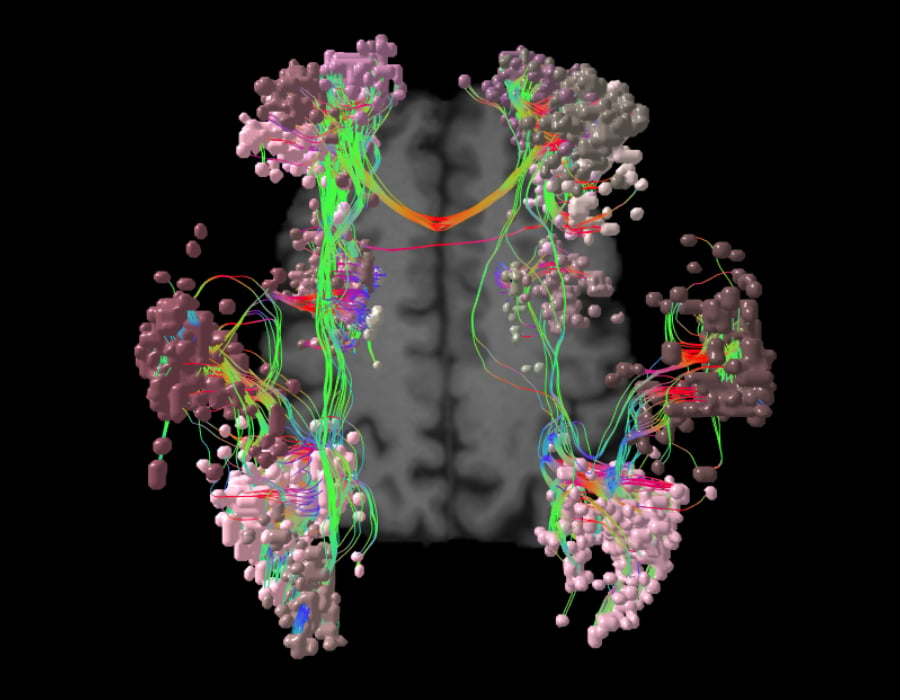
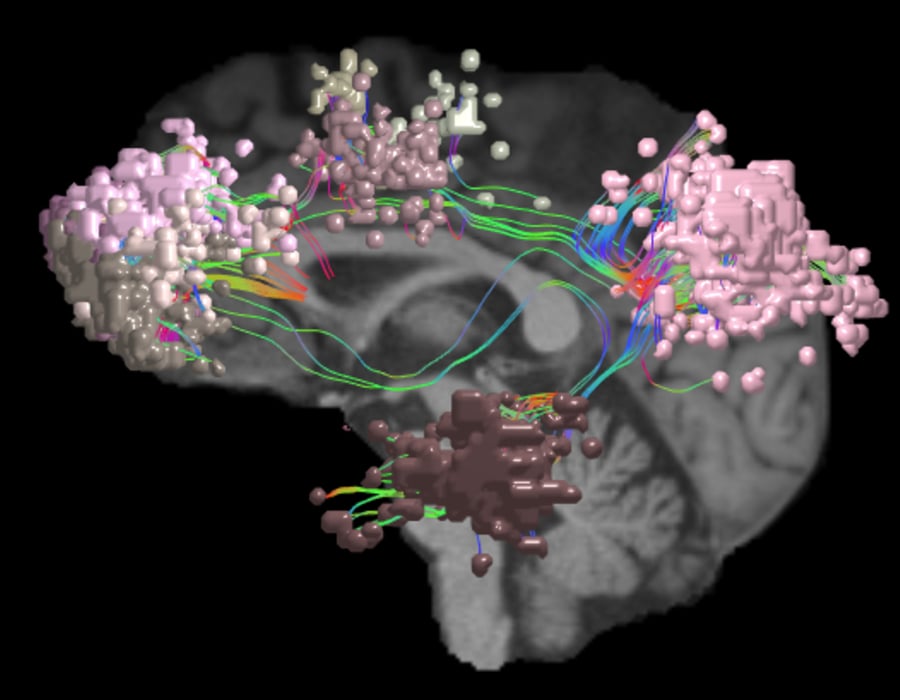
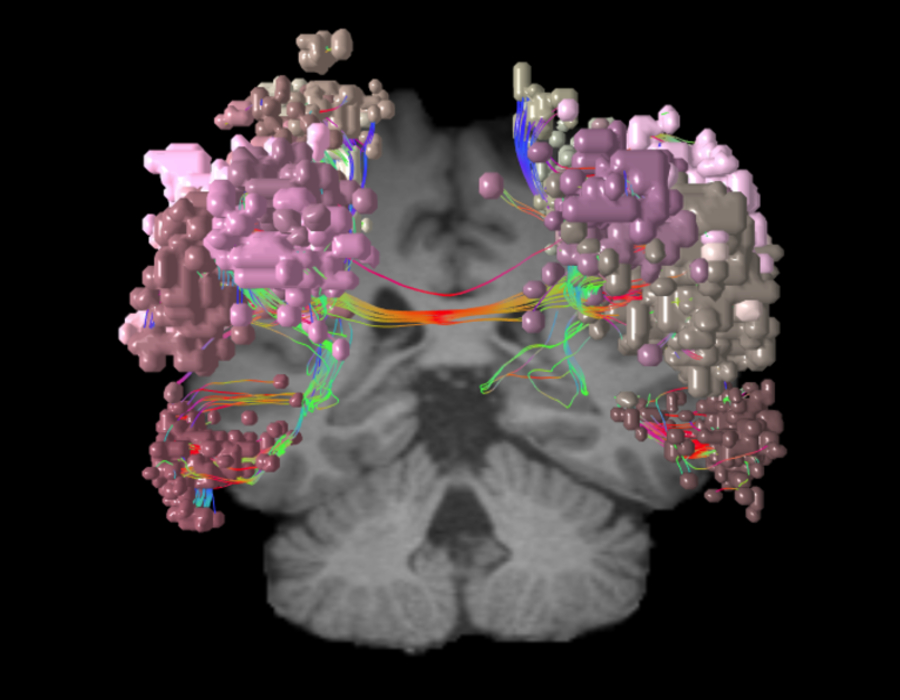
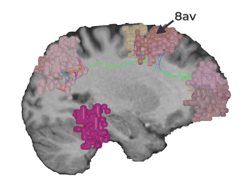
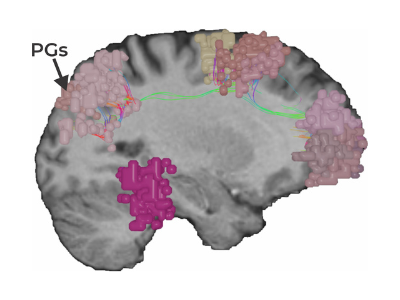
As one of the brain’s dominant control networks, the central executive network works closely with each of the brain’s other main networks.
Like previously mentioned, the CEN correlates with the DAN for attention processing, as well as visual spatial planning.8
The CEN’s external mind is also anticorrelated with the DMN’s internal mind, which focuses on reflective, task-negative processes.18,19In healthy brains, the CEN and DMN have been shown to specifically alternate activity, forming a pair of anticorrelated networks.3 When the CEN is active, such as when processing visual or sensory inputs for task selection, the DMN is inactive; conversely, when the DMN is activated, such as during contemplation or daydreaming, the CEN is deactivated.20
The salience network (SN) is responsible for the switch between the CEN and DMN for external and internal processing.21
To return to our kitchen analogy, if you were hungry but decided you were not interested in eating either an apple or cookies, you could make a sandwich. The CEN begins issuing directives to locate the items you would need: a plate, bread, deli meat, and condiments.
Once you have assembled the sandwich and started eating, active task processing is no longer required. Your mind can then start to wander. If you begin thinking about your plans for the weekend or recalling something that happened earlier in the day, the salience network has imperceptibly switched your brain’s dominant control from the CEN to the DMN.
In addition to its relationship with the SN and DMN, the central executive network also integrates with each of the other networks as needed for task processing. The CEN:
- Receives visual inputs from the visual network
- Evaluates auditory inputs from the auditory network
- Receives sensory inputs from the sensorimotor network while also sending back task directives8
- Processes stimuli and motivational signals from the limbic system