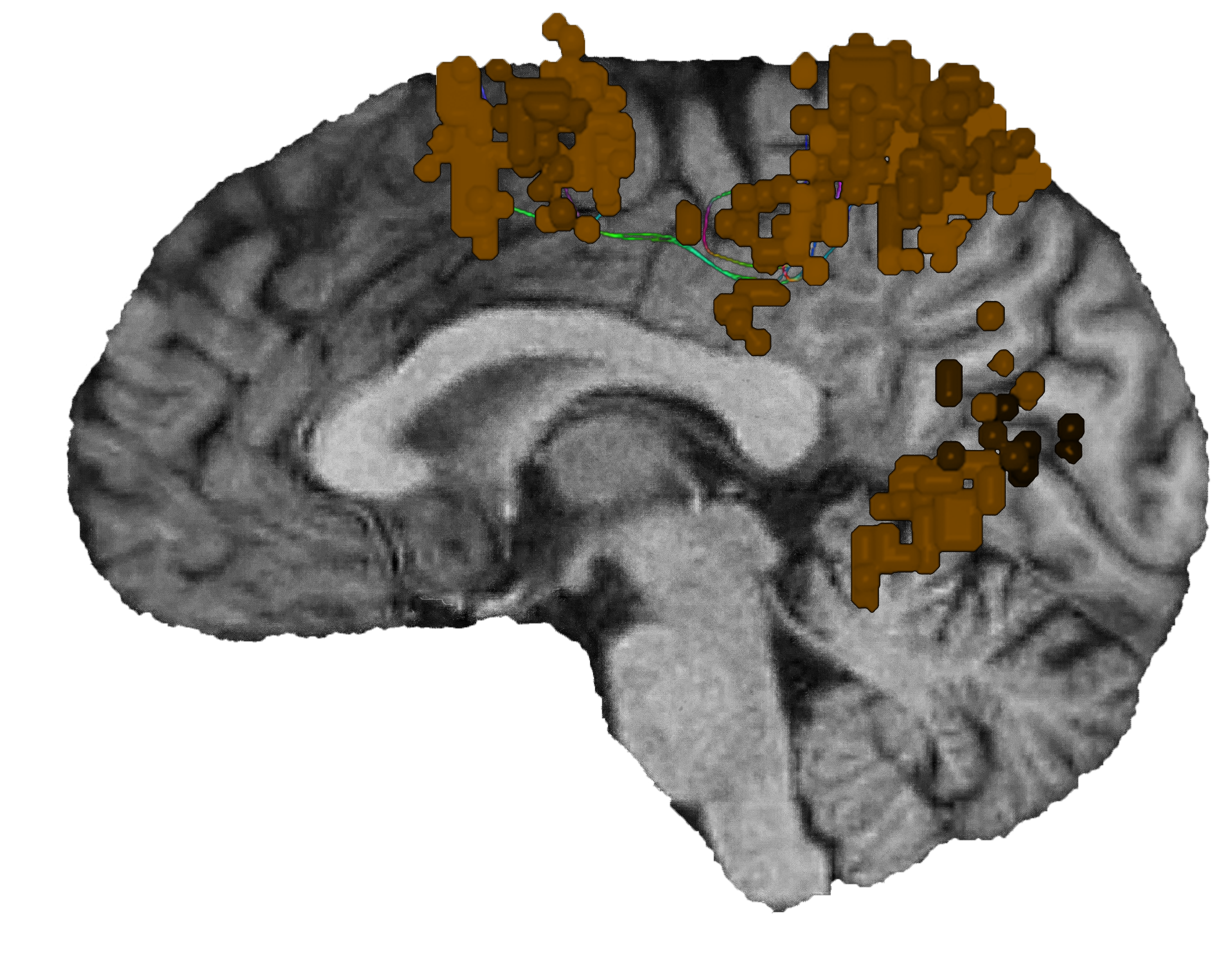
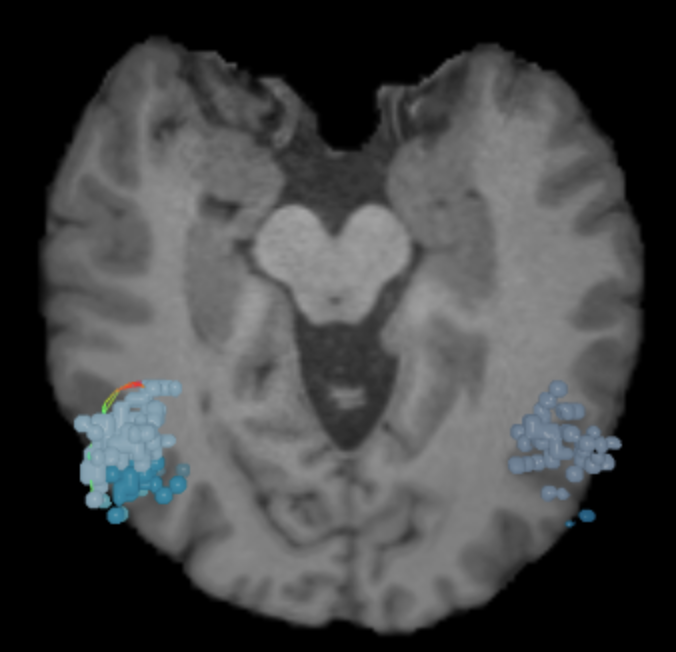
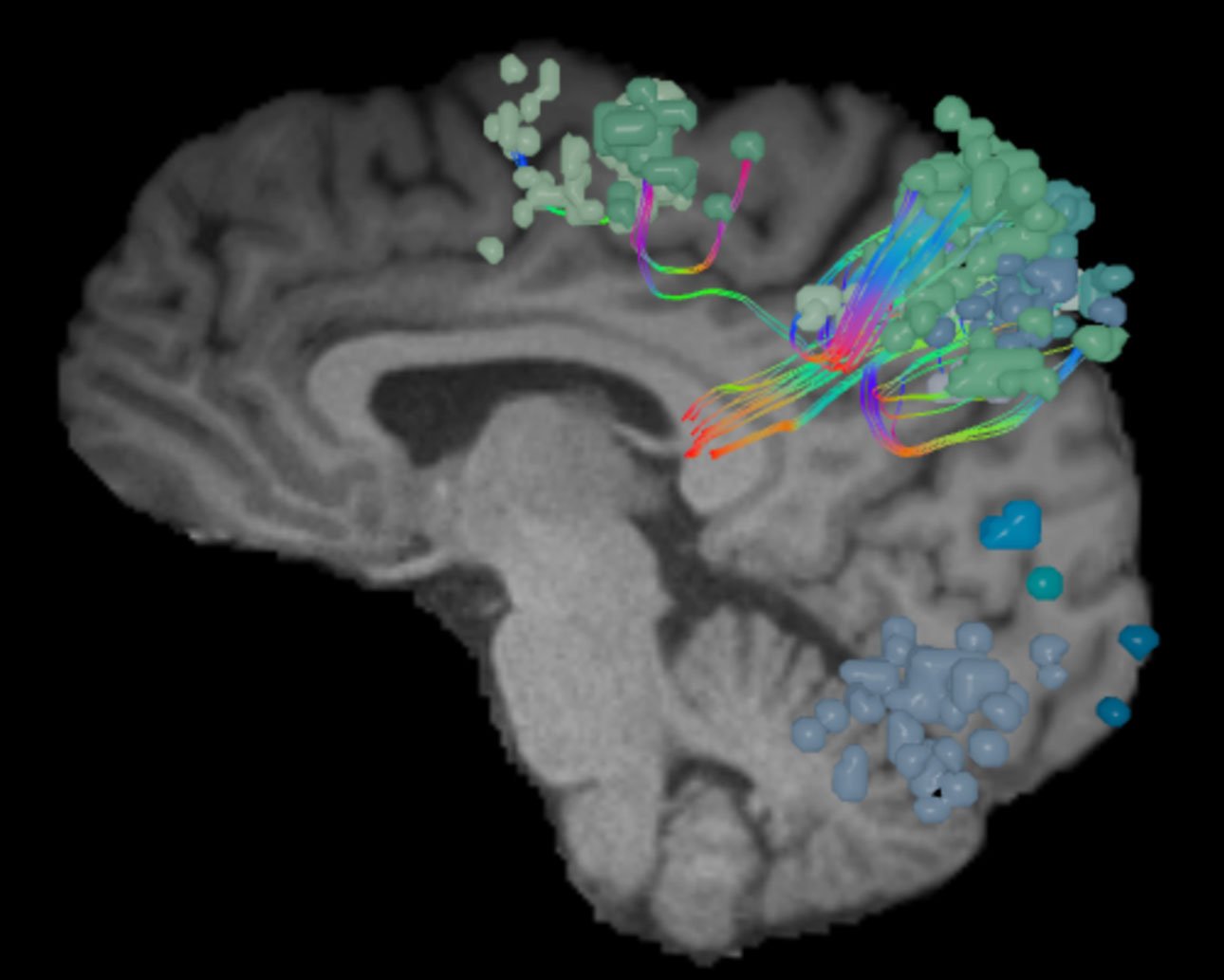
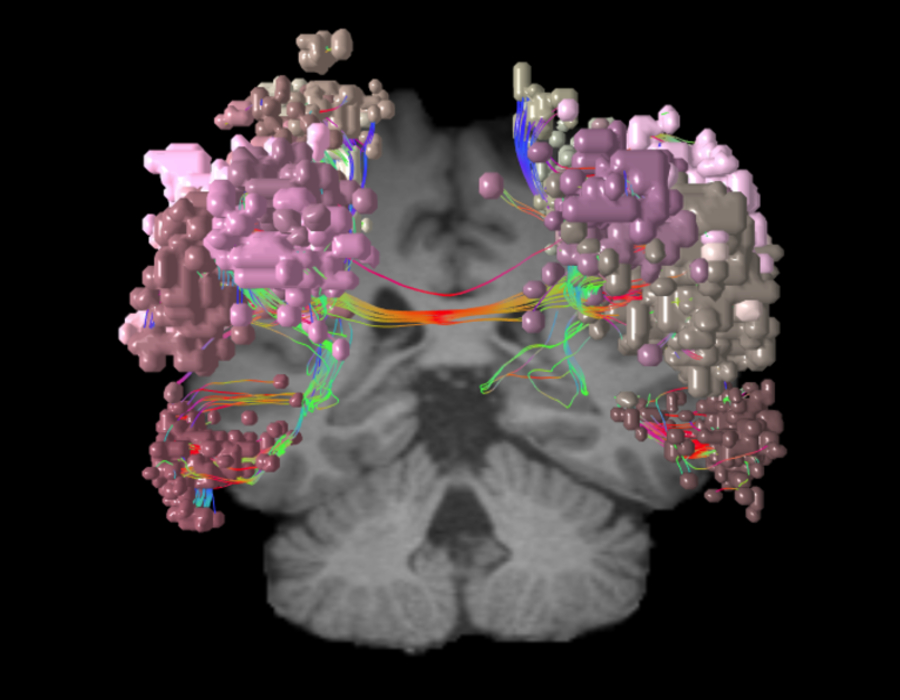
What is the dorsal attention network?
As the network that focuses human attention, the dorsal attention network (DAN) is the brain’s “aperture.”
The DAN is a consistent, bilateral network for the steady holding of attention, and it has many ways of contributing to intellectual capabilities.
As the brain’s aperture, the DAN is frequently activated in relation to other active brain networks. To illustrate these relationships, think of attention as a camera lens. Imagine photographing a garden, with each of the main brain networks as a different flower. When shifting the camera from one to the next, the aperture is adjusted to bring each new plant into sharp focus. The DAN responds similarly, guiding attention to whichever network (or networks) is most prominent and active.
The human brain receives sensory input constantly, and it cannot equally attend to every sensory signal at all times. Instead, the DAN focuses the brain’s attention to hone in on a particular moment’s most important sensory inputs.
To return to the analogy, as you decide how to frame the camera’s shot, your attention is focused on the visual elements in front of you. Though your sensorimotor network is still actively sending sensory cues about the feel of the camera and the heat of the sun, these are not your primary focus because your attention is on the photograph. The DAN selects the most pertinent sensory information for a particular moment to guide your attention.