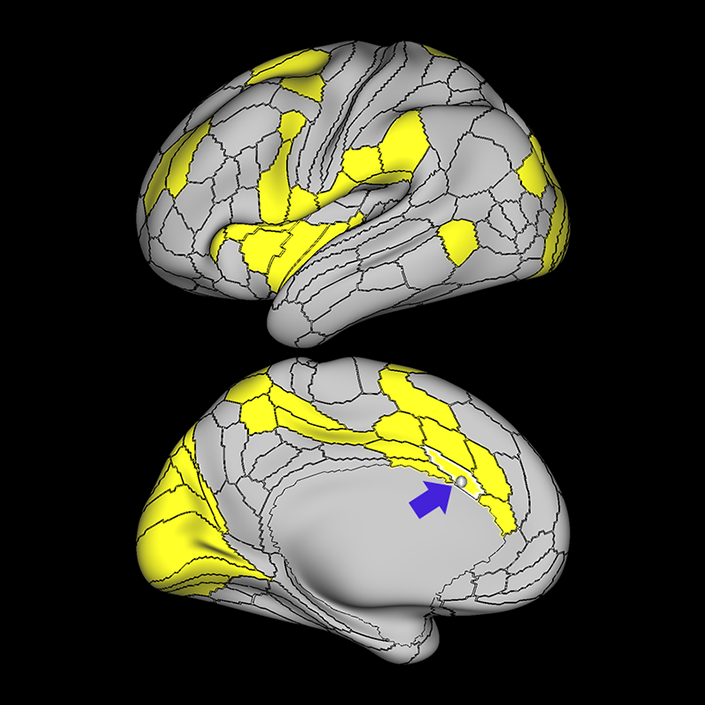
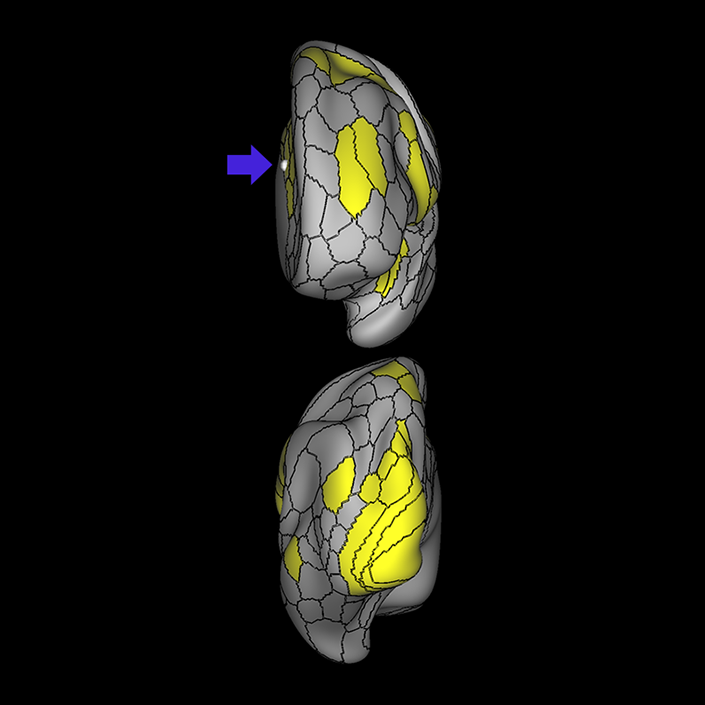
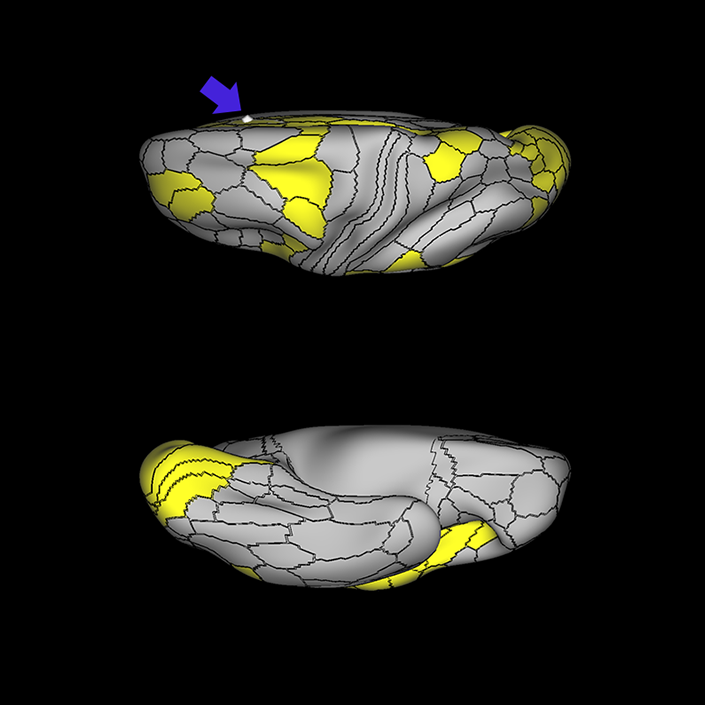
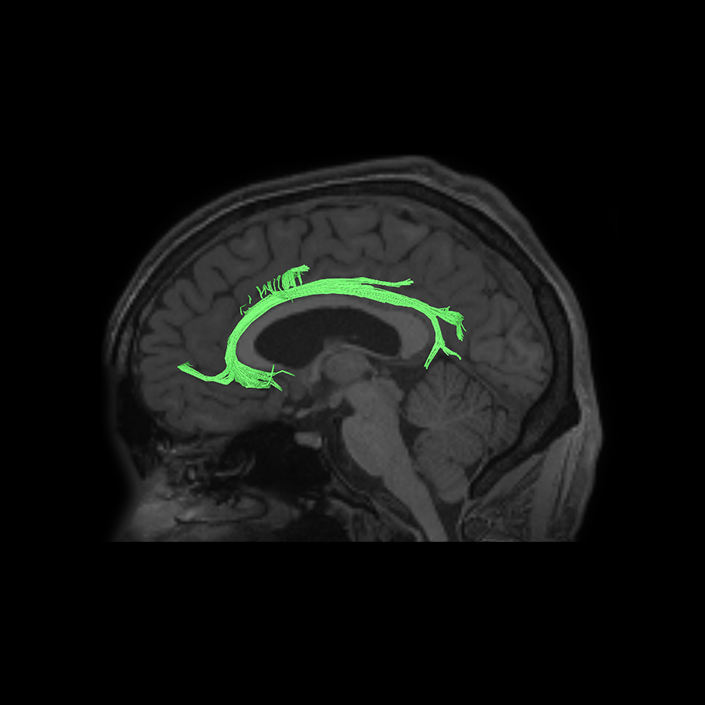
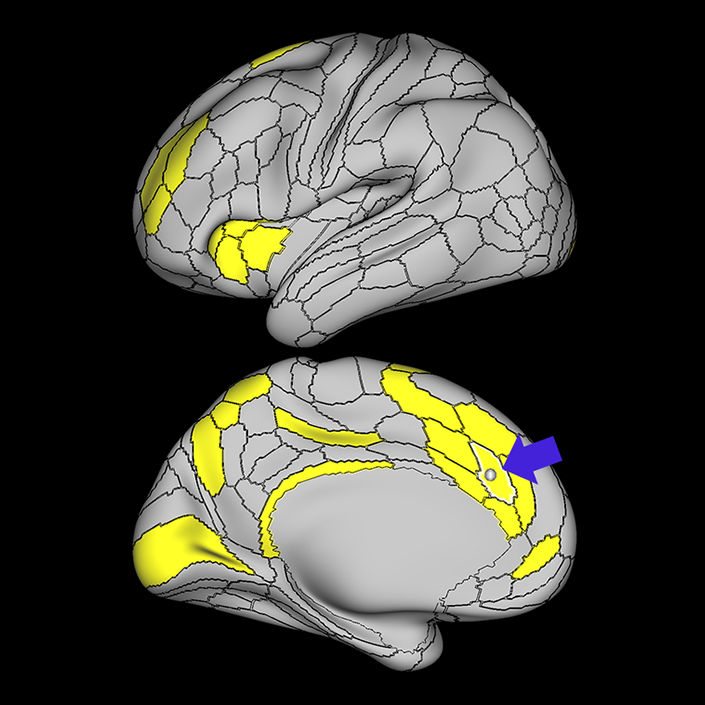
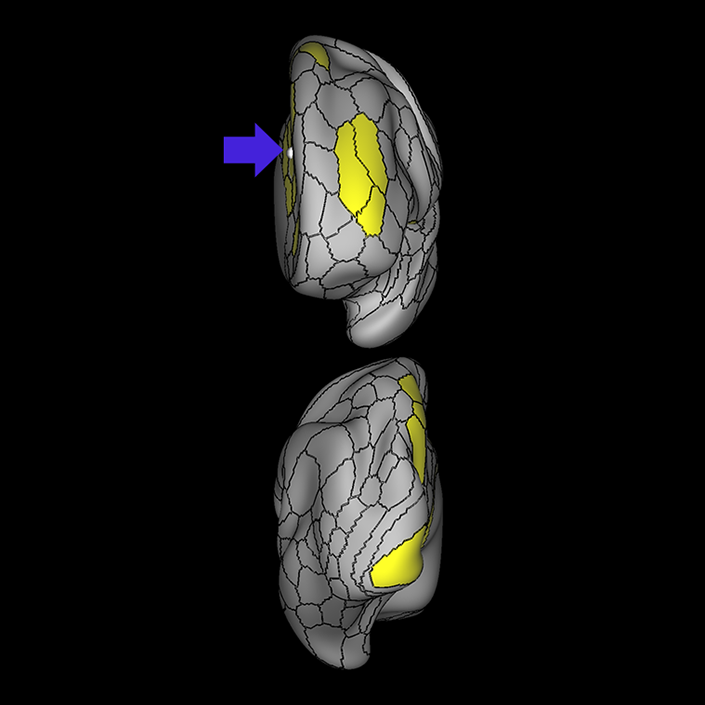
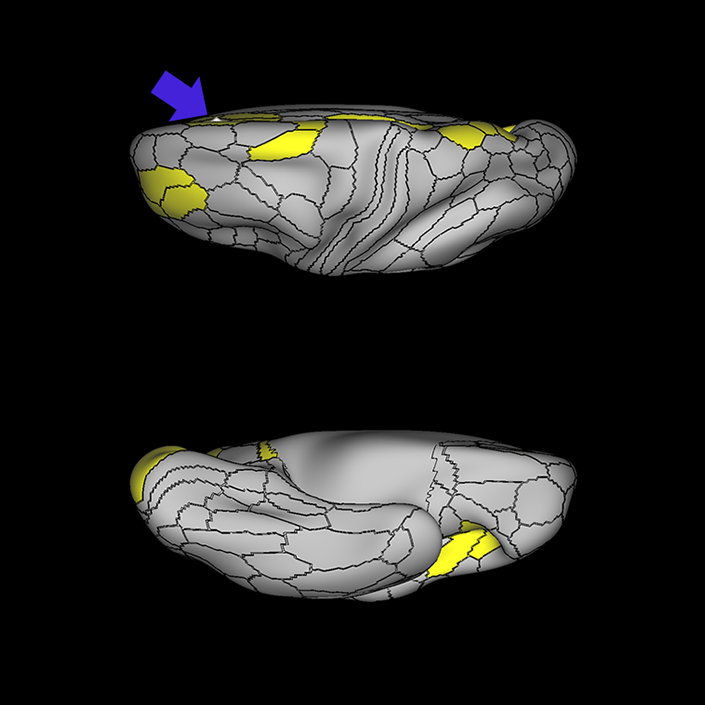
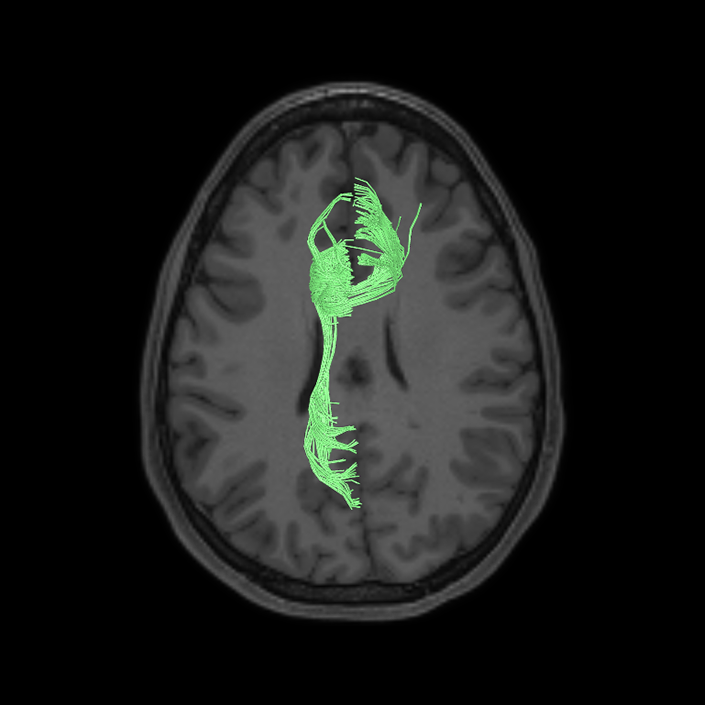
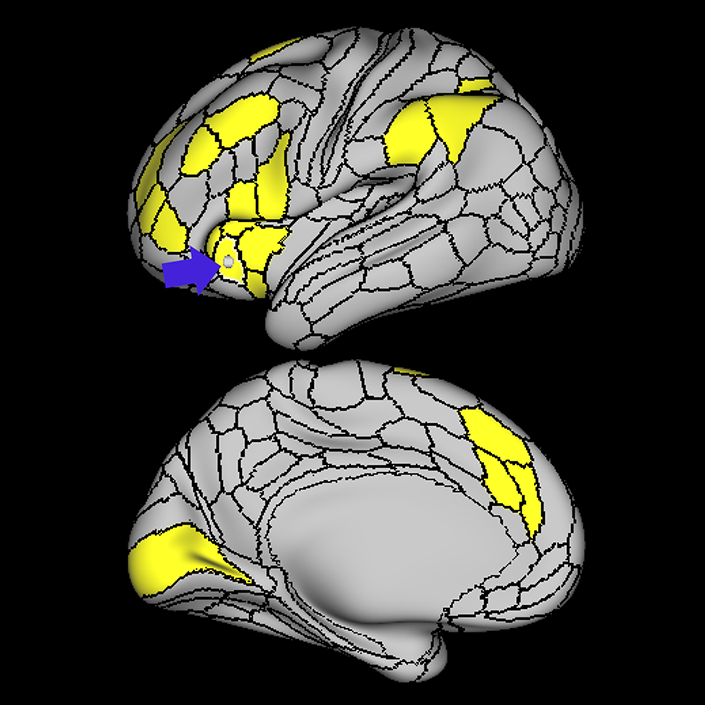
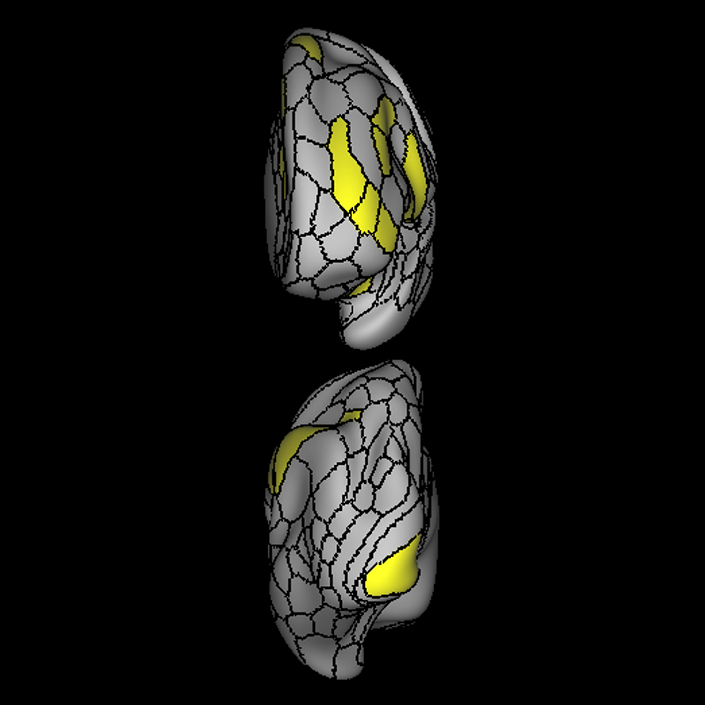
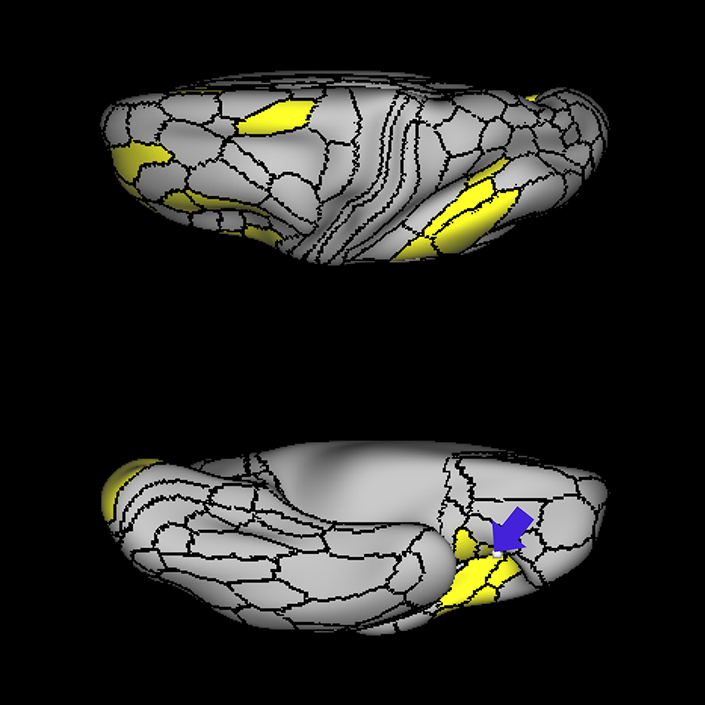
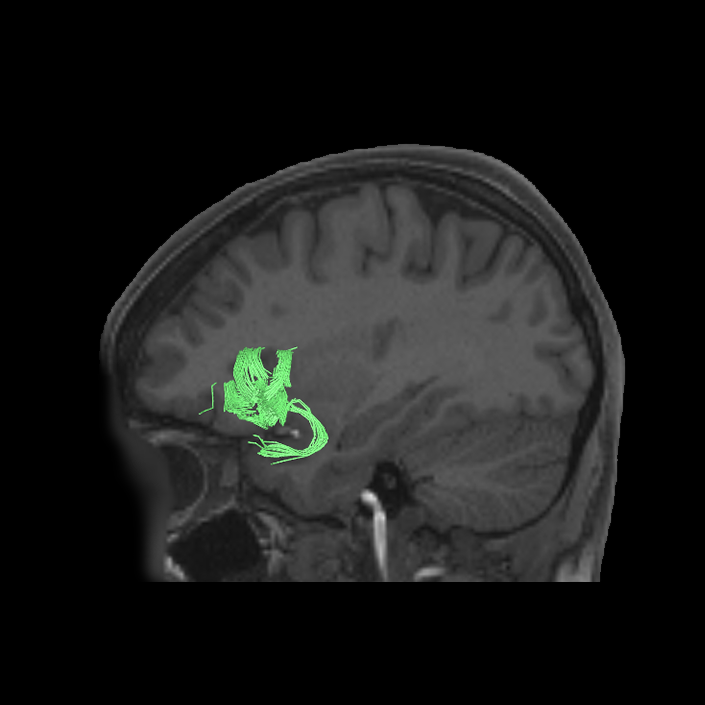
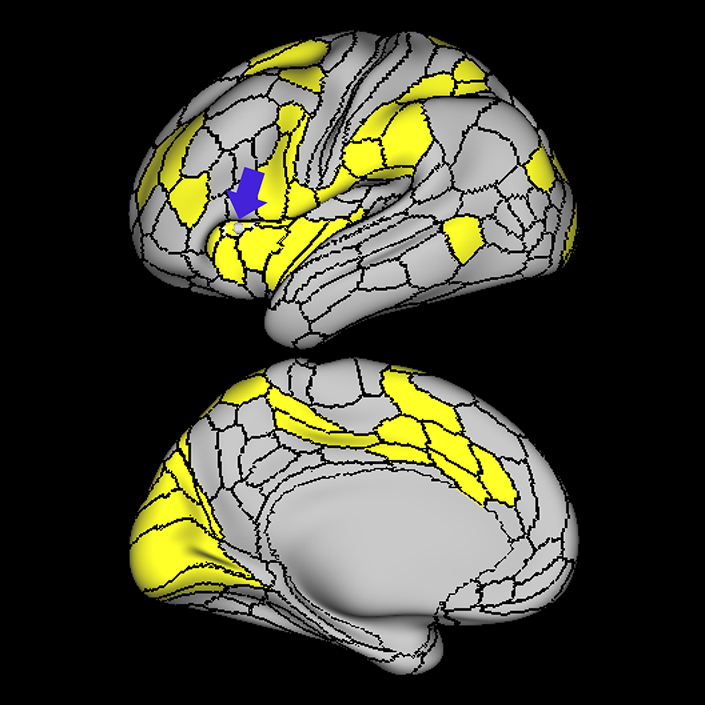
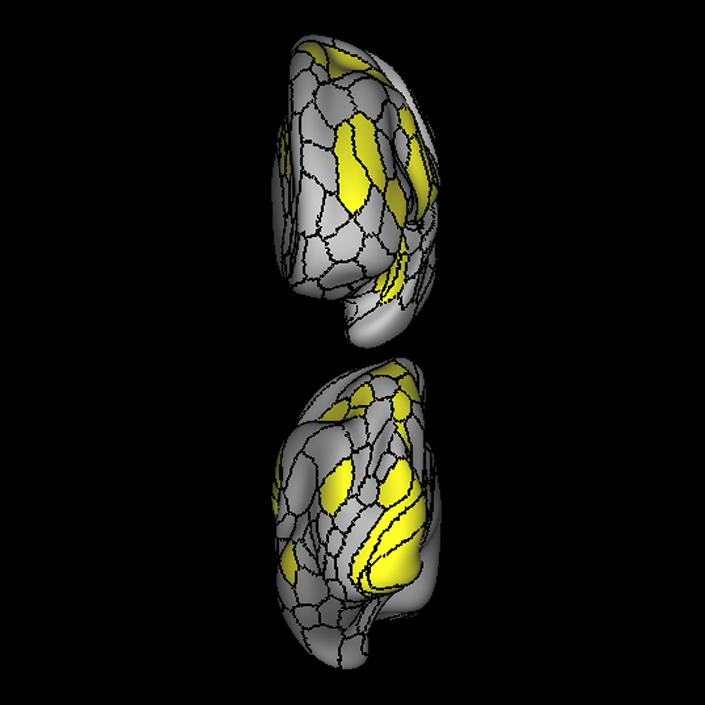
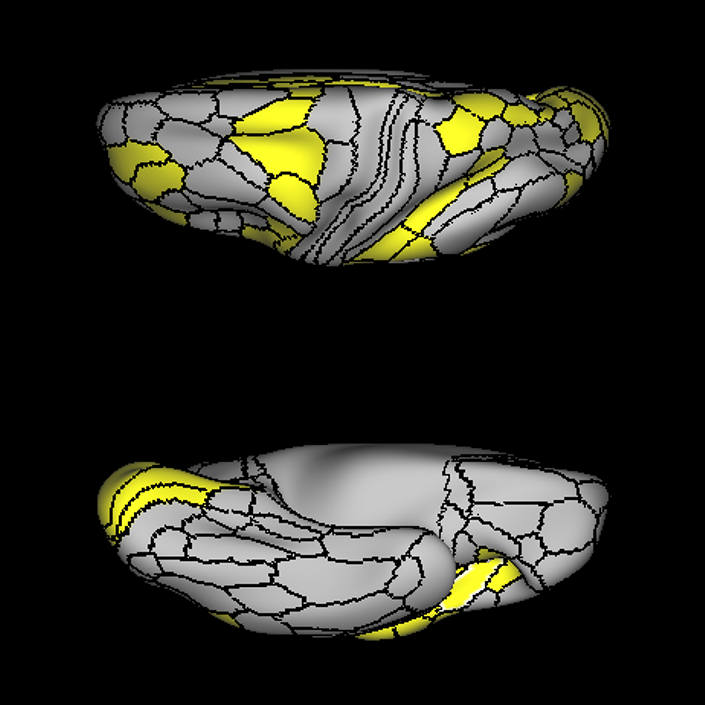
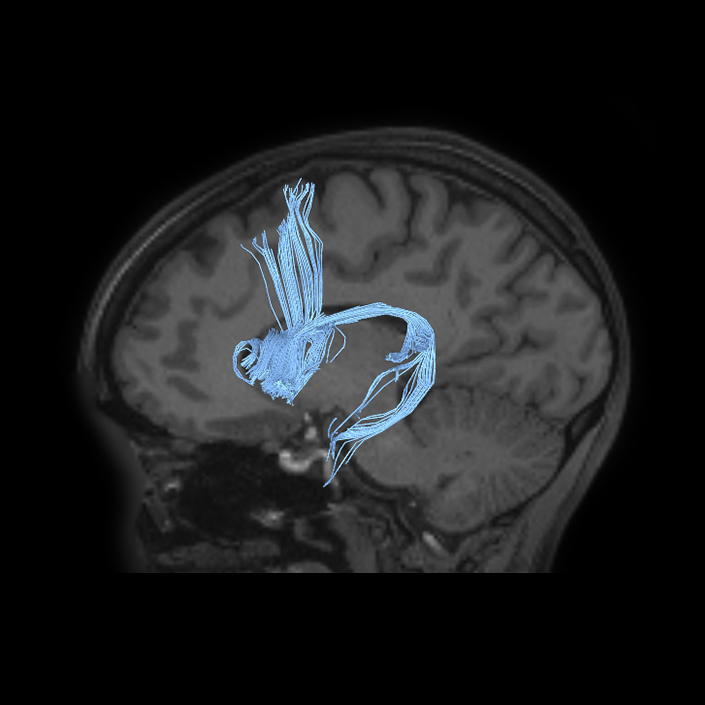
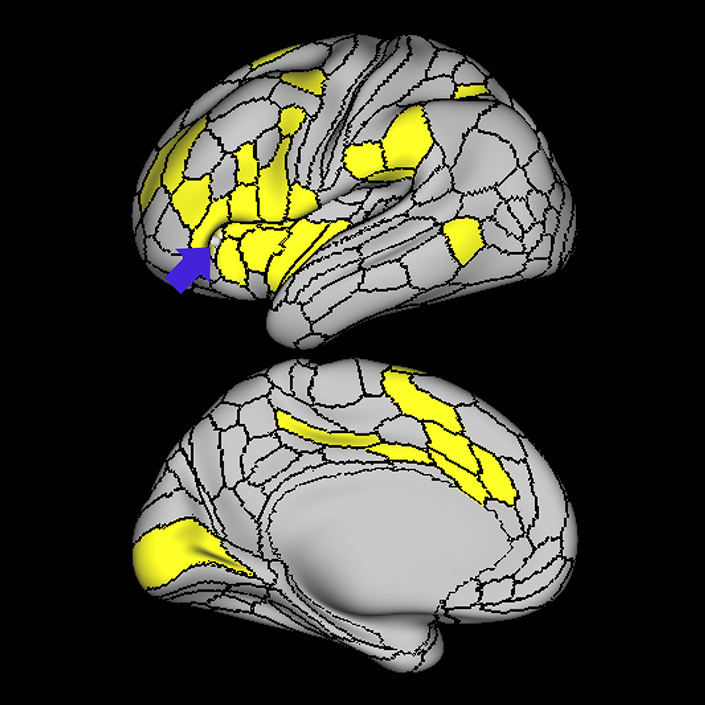
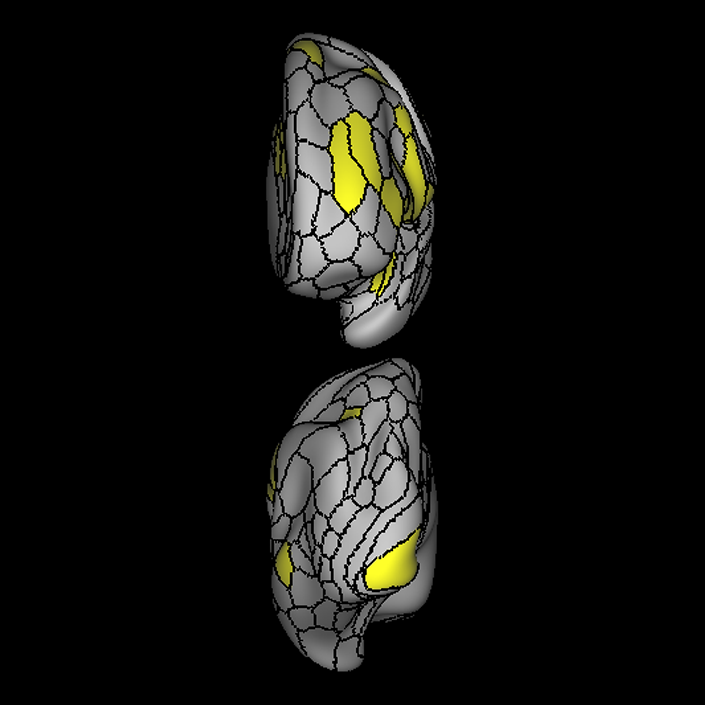
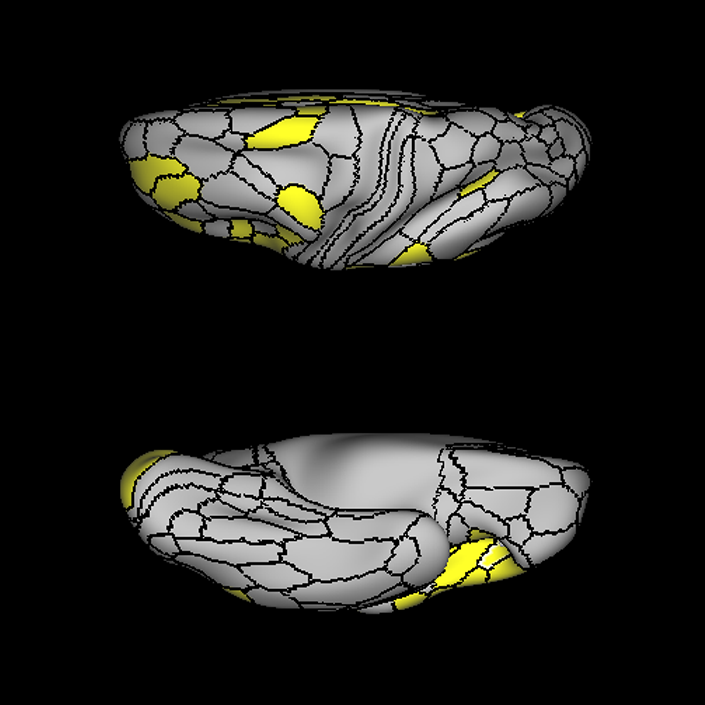
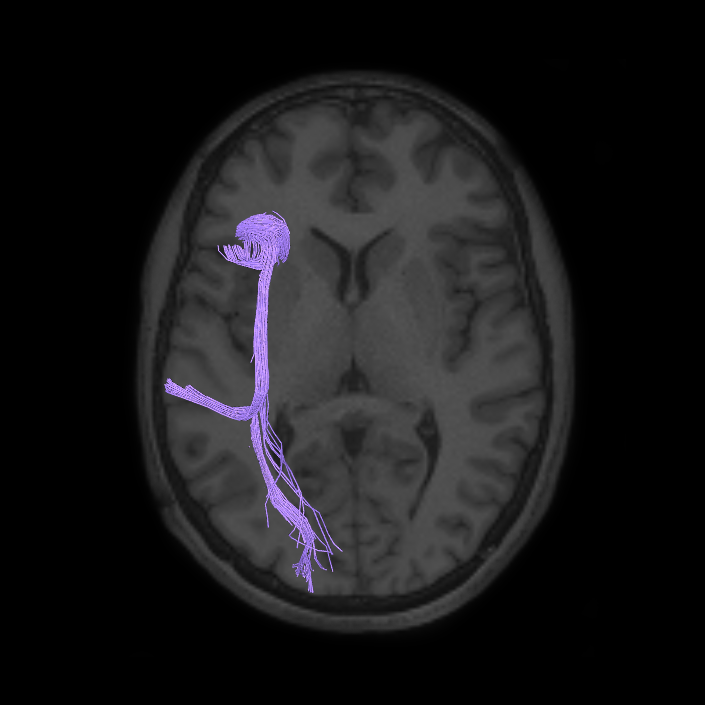
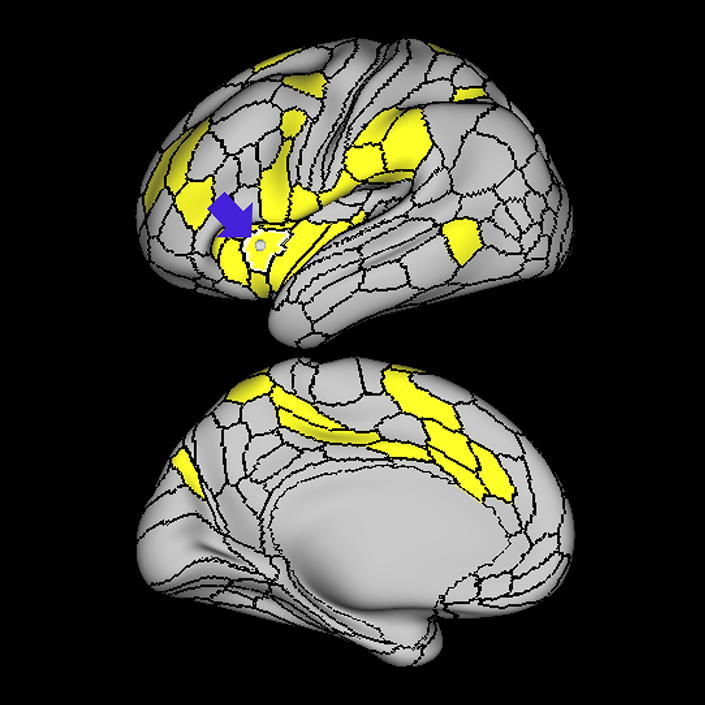
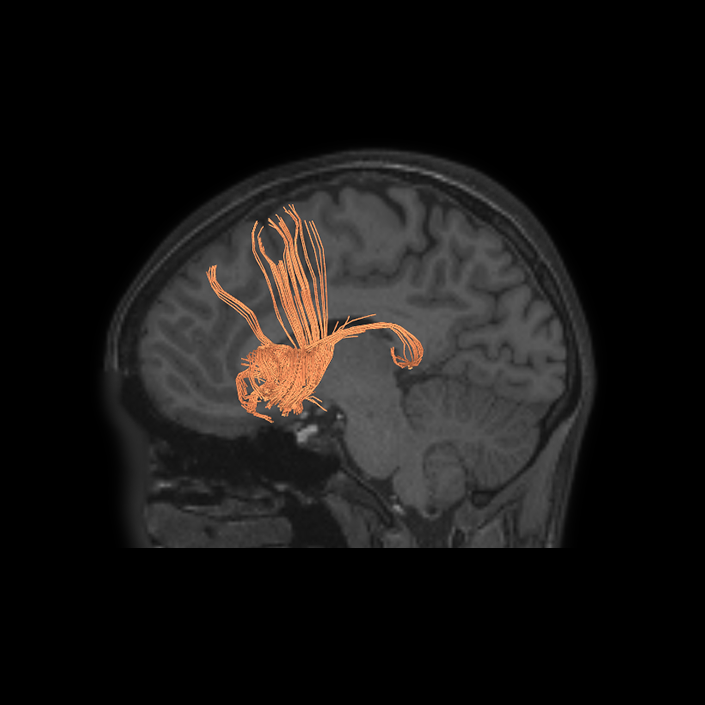
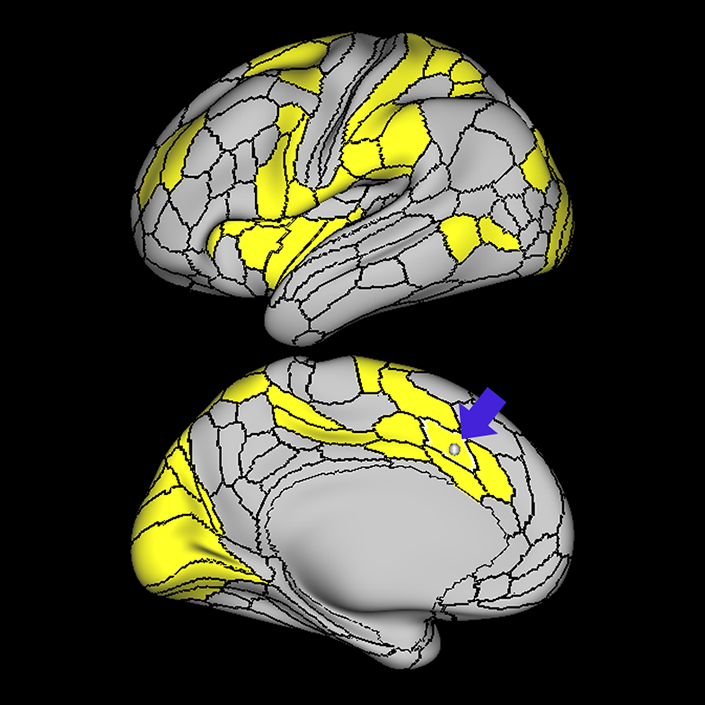
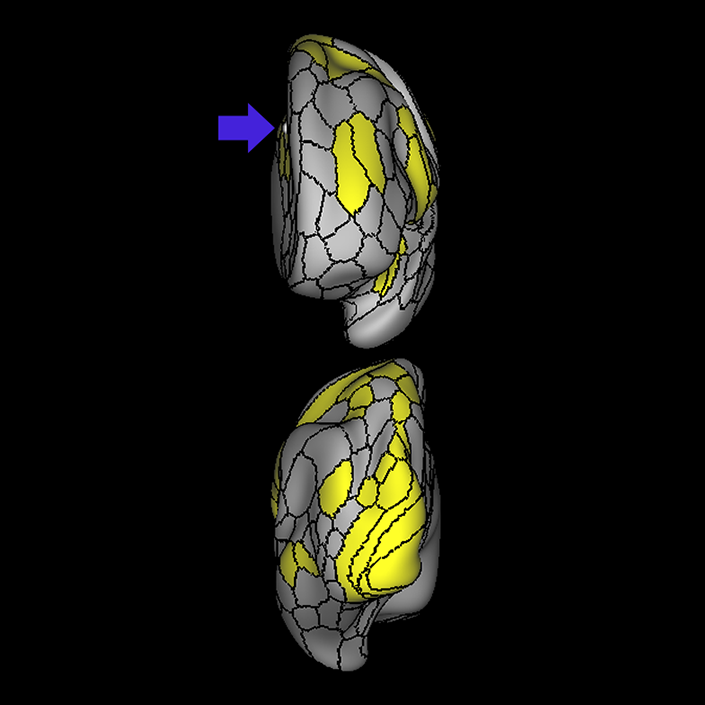
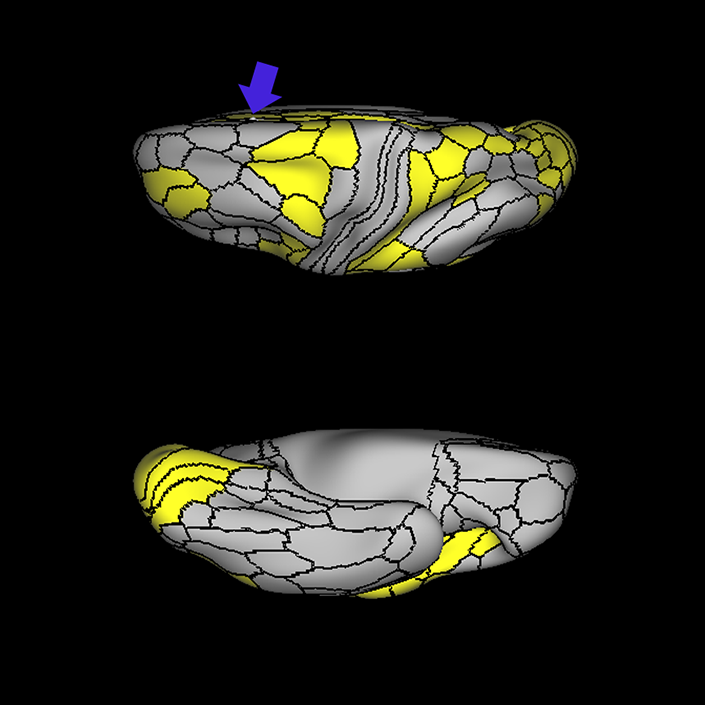
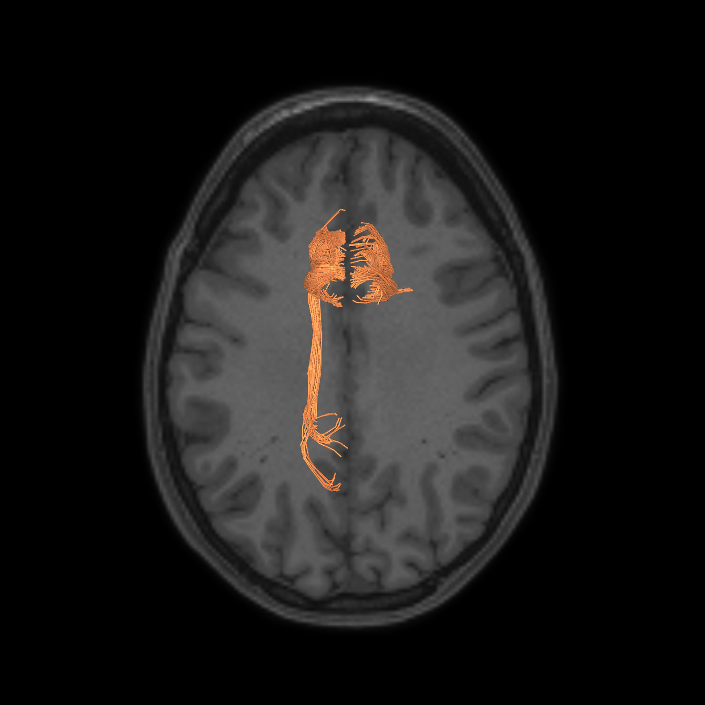
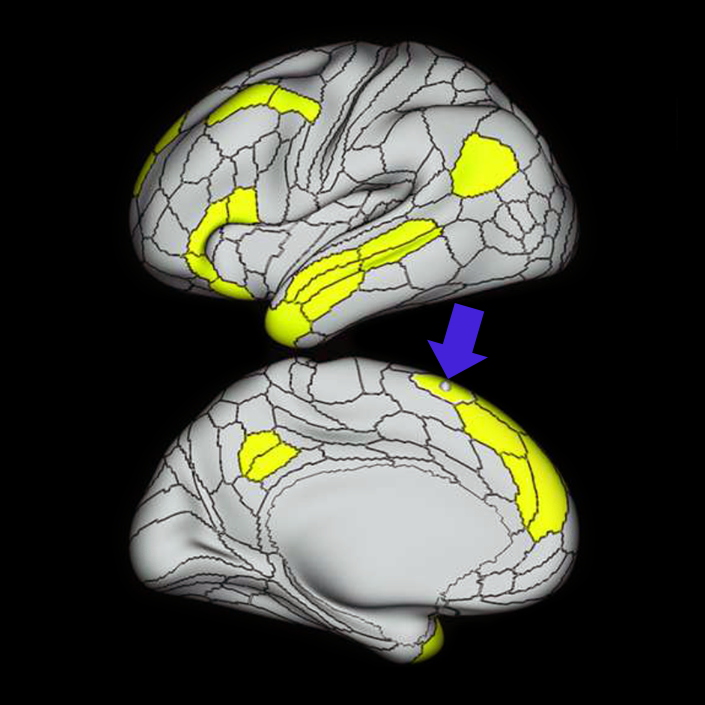
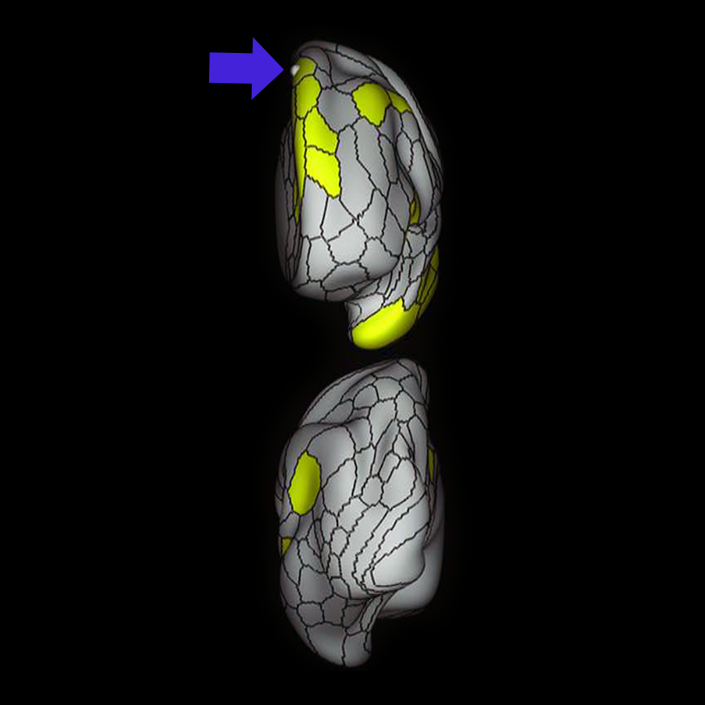
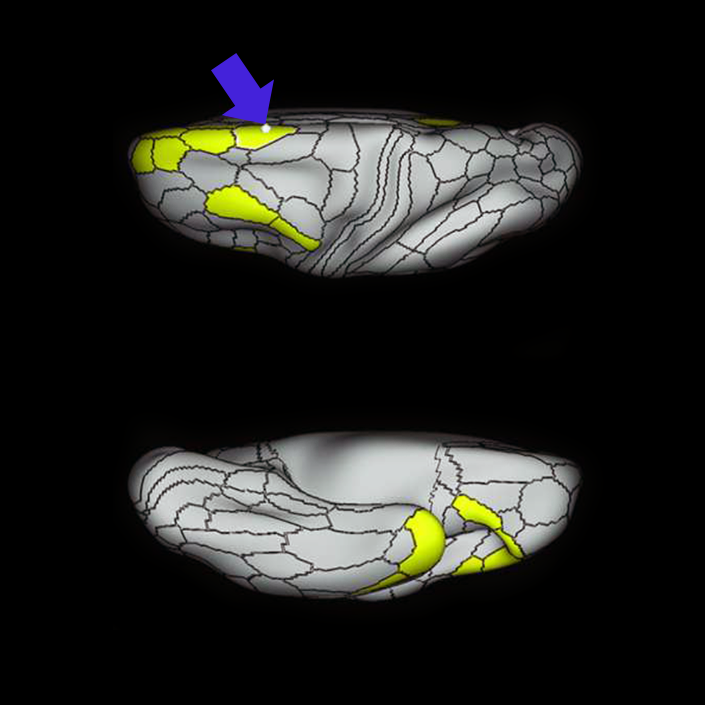
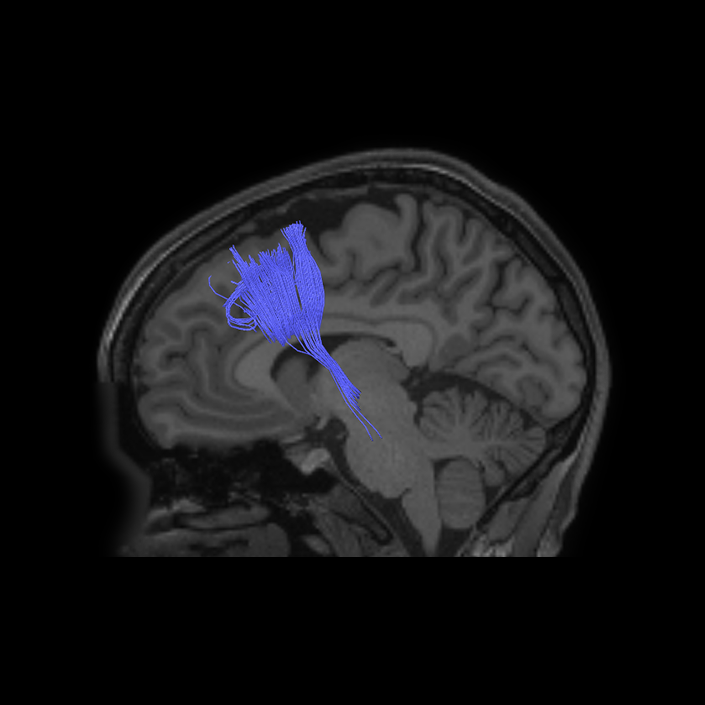
ᐅ SummaryArea a24pr (anterior 24prime): part of anterior cingulate regions. Shows little response to affective processes, but is implicated in cognitive response selection, and has been implicated in word and sentence selection during language-based tasks. ᐅ Where is it?Area a24pr (anterior 24prime) is located in the middle cingulate gyrus. It is primarily located in the superior half of the gyrus, and straddles into the inferior bank of the cingulate sulcus. ᐅ What are its borders?Area a24pr borders area 33prime inferiorly, p24pr posteriorly, p24 anteriorly and it shares its portion of the cingulate with p32pr superiorly. ᐅ What are its functional connections?Area a24pr is connected to areas 33prime, 5mv, 23d, 24dv, p24, p24prime, and a32prime, and p32prime in the cingulate areas, areas SCEF, FEF, PEF, 6a and 6r in the premotor areas, area 9-46d, 46 in the lateral frontal lobe, areas FOP1, FOP3, FOP4, FOP5, OP4, PFcm, MI, 43, 52, PoI2, and PoI1 in the insula opercular areas, area PHT in the temporal lobe, areas PGp, PF, PFop, and 7AL in the lateral parietal lobe, areas DVT, 7am, and PCV in the medial parietal lobe, areas V1, V2, V3, and V4 in the medial occipital lobe, and areas V6, V6a, V3a, V3b, and V7 in the dorsal visual stream areas. ᐅ What are its white matter connections?Area a24pr is structurally connected to the cingulum. Some individuals have contralateral connections through the body of the corpus callosum but this tract is inconsistent. Fibers project anteriorly above the corpus callosum to end at p32, anterior cingulum fibers also curve around the rostrum of the corpus callosum to end at 25. ᐅ What is known about its function?he precuneus to end at areas POS1 and 31pv, posterior fibers also curve around the splenium of the corpus callosum to end at RSC. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
Connectome Guide
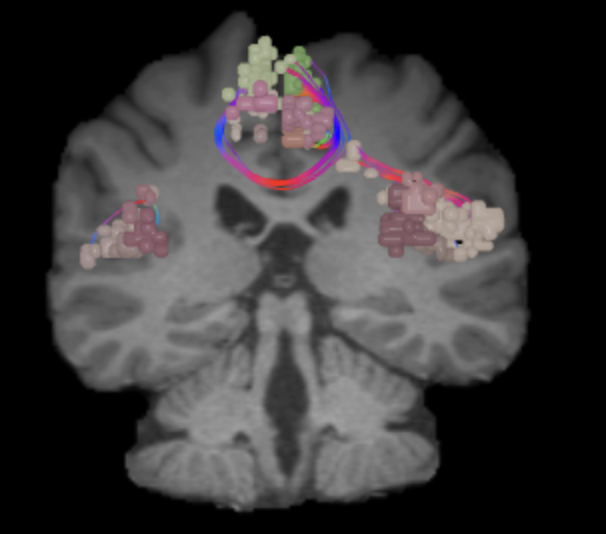
Salience Network (SN)
Why we think this network is worth considering in decision-making:
The salience network plays a key role in a number of important cognitive, emotional, and motivational function.
Evidence that this network is responsible for useful function in humans:
SN’s main components are insula and anterior cingulate cortex with three key subcortical structures, the amygdala, the ventral striatum, and the substantia nigra/ventral tegmental area.1 The anatomical connections between the components of SN in cortical level are well established in humans using diffusion tensor imaging.2,3 In addition, the neural architecture of the two cortical components of the SN shares distinct characteristics (e.g. von Economo neurons).4 Also, developmental differences have been shown both in the structural and functional connectivity level in the cortical components of SN, which was suggested to account for the cognitive maturation with age.5 SN mainly supports detecting salient stimulus to recruit functional networks while between activation and deactivation of DMN and CEN.6
Consequences of damage to this network:
Akinetic Mutism: Using lesion network mapping, Darby et (2019) have shown that lesions in the SN network cause disruptions in volition and cause akinetic mutism or abulia.7 This finding was also consistent with prior studies of disorders of free will perception, such as PNES or catatonia that identified structural and functional abnormalities in the same network. Disruptions to this network for depression, obsessive-compulsive disorder, or chronic pain introduced impairments in volition as well.8,9,10
Dementia: It has been shown that SN connectivity pattern can successfully predict different dementia types such that decreased connectivity in the SN correlated with the behavioral variant frontotemporal dementia whereas increased connectivity with the Alzheimer’s Disease. Moreover, in the behavioral variant frontotemporal dementia group, clinical severity positively correlated with the loss of SN connectivity.11
Schizophrenia: SN abnormalities are found in schizophrenia12 and correlated with the severity of reality distortion.13 Findings also highlights the importance of the SN in regulating network switching and the role of these processes in higher cognition.14 In addition to functional connectivity impairments, studies found volumetric grey matter changes in SN regions in patients with schizophrenia.15
ADHD: Additional studies identified lower connectivity in the anatomical correlates of SN in ADHD.16 Using machine-learning algorithms, Sato et al could establish an abnormality matrix derived from the connectivity patterns of SD regions, and this matrix could successfully classify adults with ADHD.17
Other Mental Disorders: In obsessive-compulsive disorder, abnormalities in salience network connectivity measures related to symptom severity as well as decreased sustained attention.18 Impairments in connectivity in SN associated with higher trait anxiety,19 ruminative brooding20 and depression.21 Volumetric grey matter changes in SD also identified in depression patients.14
ᐅ SummaryArea a32pr (anterior 32 prime): part of anterior cingulate regions. Research suggests that a32pr helps to guide behavior by evaluating motivation, anticipating outcomes, recognizing reward values, and encoding errors to influence attention allocation and motor preparation. ᐅ Where is it?Area a32pr (anterior 32 prime) is located in the posterior inferior portion of the superior frontal gyrus. It wraps into the superior bank of the cingulate sulcus. ᐅ What are its borders?Area a32pr borders area 8BM superiorly, p32pr posteriorly, a24pr and p24 inferiorly, and d32 anteriorly. ᐅ What are its functional connections?Area a32pr demonstrates functional connectivity to areas 8BM, SCEF, p24, d32, 23c, p24prime, and p32prime in the medial frontal lobe, areas a9-46v, 9- 46d, and 46 in the lateral frontal lobe, areas FOP4, FOP5, AVI and MI in the insula opercular areas, areas 7pm, RSC and POS2 in the medial parietal lobe, area V1 in the medial occipital lobe. ᐅ What are its white matter connections?Area a32pr is structurally connected to the cingulum and contralateral hemisphere. Contralateral connections course through the body of the corpus callosum to end at a32pr, p32pr, 8BM and 9m. Cingulum fibers project posteriorly from a32pr to end at precuneus areas 7m, 31a, 31pd, 31pv, RSC, Sand v23ab. Local short association fibers connect with p24, 8BM, SCEF and d32. ᐅ What is known about its function?Research on this particular region suggests that a32pr helps to guide behavior by evaluating motivation, anticipating outcomes, recognizing reward values, and encoding errors to influence attention allocation and motor preparation. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea AVI (anterior ventral insula): part of anterior apex regions. Suggested to have a role in sensation and control of autonomic nervous system processes as well as playing a role in human awareness, self-recognition, time perception, and perceptual decision making. ᐅ Where is it?Area AVI (anterior ventral insula) is located in anterior superior apex of the insula. ᐅ What are its borders?Area AVI borders area 47l anteriorly, areas 47s and AAIC inferiorly, MI posteriorly, and FOP4 and FOP5 superiorly. ᐅ What are its functional connections?Area AVI demonstrates functional connectivity to areas 6ma and 6r in the premotor regions, areas 44, p47r, 8C, a9-46v, p9-46v and 9-46d in the lateral frontal lobe, areas 8BM d32, and a32prime in the medial frontal lobe, areas FOP4, and FOP5 in the superior insula opercular regions, areas AAIC and MI in the lower opercula and Heschl's gyrus regions, areas TE2p, PHA3 and PHT in the temporal lobe, areas LIPd, PF, PFm, and IP2 in the lateral parietal lobe, area V1 in the medial occipital lobe. ᐅ What are its white matter connections?Area AVI is structurally connected to local parcellations. Local short association bundles connect to insular parcellations 47s, 47l, AAIC, FOP4, FOP5, and MI, and temporal pole parcellations TGd. ᐅ What is known about its function?Area AVI is a newly described area of the brain and was parcellated from the anterior insula. The anterior insula is suggested to have a role in sensation and control of autonomic nervous system processes as well as playing a role in human awareness, self-recognition, time perception, and perceptual decision making. Area AVI was parcellated from areas MI and AAIC in the anterior insula based on functional activity differences between regions related to motor, arithmetic, auditory language, and semantic tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea FOP4 (frontal operculum 4): part of anterior apex regions. The frontal operculum plays a key role in the initiation of language and lexical retrieval required for language learning. ᐅ Where is it?Area FOP4 (Frontal operculum 4) is located on the inner surface of the pars opercularis of the IFG. ᐅ What are its borders?Area FOP4 borders area 44 on its exterior surface as well as a small border with area 6r posterosuperiorly. Its posterior border is made up of FOP1 and FOP3. Its inferior border is with MI and AVI. Its anterior border is with FOP5. ᐅ What are its functional connections?Area FOP4 demonstrates functional connectivity to areas SCEF, FEF, PEF, 6v, 6a, 6ma, and 6r in the premotor regions, areas IFSa, 46, and 9-46d in the lateral frontal lobe, areas 5mv, 23c, 24dv, a24prime, p24prime, a32prime, and p32prime in the medial frontal lobe, areas 43, PFcm, OP4, FOP3, and FOP5 in the superior insula opercular regions, areas AVI, MI, PI, 52, PoI1, and PoI2 in the lower opercula and Heschl's gyrus regions, area PHT in the temporal lobe, areas 7AL, 7PL, AIP, LIPv, LIPd, PF, PFt, PGp, and PFop in the lateral parietal lobe, area V6 in the dorsal visual stream area, area 7am and DVT in the medial parietal lobe, areas V1, V2, and V3 in the medial occipital lobe. ᐅ What are its white matter connections?Area FOP4 is structurally connected to the frontal aslant tract and the arcuate/SLF. Connections from FOP4 with the frontal aslant tract project superior to the superior frontal gyrus to end at parcellations 6ma and SFL. Arcuate/SLF fibers project posteriorly above the insula, curving around the termination of the sylvain fissure to end at TGd and TE1a. From the arcuate/SLF there are also projections to the superior temporal gyrus that end at A4 and A5. Local short association fibers connect with AVI, FOP3, FOP5 and MI 14. White matter connections from FOP4 in the right hemisphere have less consistent connections with the arcuate/SLF ᐅ What is known about its function?Area FOP4 is a newly described area of the brain that was parcellated from the frontal operculum. The frontal operculum plays a key role in the initiation of language and lexical retrieval required for language learning. Area FOP4 was parcellated from areas AVI and FOP1 based on differences in functional activity in motor-based tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea FOP5 (frontal operculum 5): part of anterior apex regions. Frontal operculum plays a key role in the initiation of language and lexical retrieval required for language learning. FOP5 showed greater activity in motor tasks during which individuals squeezed their left and right toes, tapped their left and right fingers, and moved their tongue. ᐅ Where is it?Area FOP5 (frontal operculum 5) is located on the undersurface of the opercular portions of pars triangularis of the IFG. ᐅ What are its borders?Area FOP5 is principally the underside of area 45, though parts of area 44 and area 47L are on its exterior surface. AVI is inferior to it and FOP4 is posterior to it. ᐅ What are its functional connections?Area FOP5 demonstrates functional connectivity to areas SCEF, FEF, PEF, 6ma and 6r in the premotor regions, areas 44, 45, IFSa, IFja, 46, and 9-46d in the lateral frontal lobe, areas 23c, a24prime, p24prime, a32prime, and p32prime in the medial frontal lobe, areas 43, PFcm, FOP3, and FOP4 in the superior insula opercular regions, areas AVI, MI, PoI1, and PoI2 in the lower opercula and Heschl's gyrus regions, area PHT in the temporal lobe, areas LIPd, PF, and PFop in the lateral parietal lobe, area 7am in the medial parietal lobe, area V1 in the medial occipital lobe. ᐅ What are its white matter connections?Area FOP5 is structurally connected to the IFOF and arcuate/SLF. The IFOF courses from occipital lobe parcellations V1, V2 V3, and parietal area MIP, through the extreme/external capsule to turn laterally to FOP5. FOP5 has arcuate/SLF fibers projecting posteriorly above the insula and turning laterally to end at the posterior insula and planum temporale parcellations RI and LBelt. Local short association fibers connect with 45, FOP4 and AVI. White matter tracts from FOP5 in the right hemisphere have less consistent connections with the arcuate/SLF. ᐅ What is known about its function?Area FOP5 is a newly described area of the brain that was parcellated from the frontal operculum. The frontal operculum plays a key role in the initiation of language and lexical retrieval required for language learning. Area FOP5 was parcellated from FOP4 based on functional activity differences between these two regions. Specifically, FOP5 showed greater activity in motor tasks during which individuals squeezed their left and righ toes, tapped their left and right fingers, and moved their tongue. Area FOP5 was parcellated from area 44 based on functional activity differences in motor cue and semantic tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea MI (middle insula): part of anterior apex regions. Suggested to have a role in sensation and control of autonomic nervous system processes as well as playing a role in human awareness, self-recognition, time perception, and perceptual decision making. ᐅ Where is it?Area MI (Middle insula) is located in the posterior superior portion of the short insular gyrus. ᐅ What are its borders?Area MI borders AVI anteriorly, POI2 posteriorly, AAIC inferiorly and FOP3 and FOP4 superiorly. ᐅ What are its functional connections?Area MI demonstrates functional connectivity to areas SCEF, FEF, PEF, 6ma and 6r in the premotor regions, areas IFSa, 46, and 9-46d in the lateral frontal lobe, areas 5mv, 23c, a24prime, p24prime, a32prime, and p32prime in the medial frontal lobe, areas OP4, 43, PFcm, FOP1, FOP3, FOP4, and FOP5 in the superior insula opercular regions, areas 52, PI, AVI, AAIC, PoI1, and PoI2 in the lower opercula and Heschl's gyrus regions, area PHT in the temporal lobe, areas LIPd, PF, PFm, PFop, and PFt in the lateral parietal lobe, area 7am in the medial parietal lobe, area V6 in the dorsal visual stream area. ᐅ What are its white matter connections?Area MI is structurally connected with the arcuate/SLF, frontal aslant tract and local parcellations. Some individuals have connections to the parietal and occipital lobe though these tracts are inconsistent. Connections to the superior frontal gyrus through the FAT project superior to 8BL, 6ma and SFL. Superior temporal gyrus connections are portions of the arcuate/SLF and course from MI, medial and superior to the insula to end laterally at A4 and A5. Local short association bundles connect with PoI1, 47s, Pir, AAIC, AVI, FOP3, FOP4 and FOP5. ᐅ What is known about its function?Area MI is a newly described area of the brain and was parcellated from the anterior insula. The anterior insula is suggested to have a role in sensation and control of autonomic nervous system processes as well as playing a role in human awareness, self-recognition, time perception, and perceptual decision making. Area MI was parcellated from areas AAIC and AVI in the anterior insula based on functional activity differences between regions related to motor, arithmetic, auditory language, and semantic tasks. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea p32pr (posterior 32 prime): part of anterior cingulate regions. Has been implicated as part of the "cognitive division" of the anterior cingulate cortex and is involved in stimulus and response selection in tasks that require attention. for linguistic and sensory information. ᐅ Where is it?Area p32pr (posterior 32 prime) is located in the posterior inferior portion of the superior frontal gyrus. It wraps into the superior bank of the cingulate sulcus. ᐅ What are its borders?Area p32pr borders SCEF superiorly, 24dv posteriorly, a24pr inferiorly, and a32pr anteriorly. ᐅ What are its functional connections?Area p32pr demonstrates functional connectivity to area 2 in the sensory strip, areas 5mv, 23d, 24dv, p24, p24prime, and a32prime, and a32prime in the cingulate areas, areas SCEF, PEF, 6a, 6v, 6ma, and 6mp in the premotor areas, area 9-46d, 46 in the lateral frontal lobe, areas FOP1, FOP3, FOP4, FOP5, OP4, PFcm, MI, 43, 52, PoI2, and PoI1 in the insula opercular areas, area PHT in the temporal lobe, areas PGp, PFt, PF, PFop, LIPd, 7PC, 7PL, and 7AL in the lateral parietal lobe, areas DVT and 7am in the medial parietal lobe, areas V1, V2, V3, and V4 in the medial occipital lobe, areas V6, V6a, V3a, V3b, and V7 in the dorsal visual stream areas, and area FST in the lateral occipital lobe. ᐅ What are its white matter connections?Area p32pr is structurally connected to the cingulum and contralateral hemisphere. Cingulum fibers project posteriorly to from p32pr to end at precuneus areas 31a, 31pd, 31pv, PCV and v23ab. Contralateral connections course through the body of the corpus callosum to end at p32pr, SCEF and a32pr. Local short association fibers connect with p32pr, SCEF and a32pr. ᐅ What is known about its function?Area p32pr has been implicated as part of the "cognitive division" of the anterior cingulate cortex and is involved in stimulus and response selection in tasks that require attention for linguistic and sensory information. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
ᐅ SummaryArea SCEF (supplementary and cingulate eye field): part of medial superior frontal gyrus regions. Higher order oculomotor center implicated in appraising all possible oculomotor behaviors to direct primary oculomotor centers in goal- directed behavior. ᐅ Where is it?Area SCEF (supplementary and cingulate eye field) is located in the posterior medial SFG. ᐅ What are its borders?Area SCEF borders area 8BM anteriorly, areas 6ma and SFL superiorly, areas 6mp and 24dd posteriorly, and areas 24dv and p32pr inferiorly. ᐅ What are its functional connections?Area SCEF demonstrates functional connectivity to areas 1, 2, 3a, 3b in the sensory strip, area 4 in the motor strip, areas PEF, FEF, 55b, 6ma, 6mp, 6a, 6r, and 6v in the premotor regions, areas a24prime, p32prime, a32prime, 5mv, and 23c in the middle cingulate regions, areas IFJa, 46, and 9-46d in the lateral frontal lobe areas OP4, OP1, PFcm, 43, FOP1, FOP2, FOP3 FOP4, and FOP5 in the superior insula opercular regions, areas PSL, 52, A4, MI, PoI1 and PoI2 in the lower opercula and Heschl's gyrus regions, area PHT in the temporal lobe, areas AIP, MIP, VIP, LIPd, LIPv, PFop, PF, PFt, IP0, IPS1, 7AL, 7PL, and 7PC, in the lateral parietal lobe, areas 7am and DVT in the medial parietal lobe, area V1, V2, V3, V4 in the medial occipital lobe, areas V3a, V3b, V6, V6a, and V7 in the dorsal visual stream, area FFC of the ventral visual stream, and areas PH, TPOJ2, LO3, MST, and FST of the lateral occipital lobe. ᐅ What are its white matter connections?Area SCEF is structurally connected to the contralateral hemisphere and thalamus. Contralateral connections course through the body of the corpus callosum to end at SCEF, 8BL, SFL and 8BM. Thalamic projections travel through the ventral thalamus to the brainstem (Figure 32). Local short association bundles connect with SF, 8BM, SFL and 8BL. ᐅ What is known about its function?Area SCEF is a higher order oculomotor center implicated in appraising all possible oculomotor behaviors to direct primary oculomotor centers in goal-directed behavior. |
|
A: lateral-medial
B: anterior-posterior
C: superior-inferior
DTI image |
Reference list
- Menon V. Salience Network Introduction and Overview. 2015. https://doi.org/10.1016/B978-0-12-397025-1.00052-X
- Uddin Q, Supekar S, Ryali S, Menon V. Dynamic reconfiguration of structural and functional connectivity across core neurocognitive brain networks with development. Journal of Neuroscience, 2011;31(50), 18578–18589. https://doi.org/10.1523/JNEUROSCI.4465-11.2011
- van den Heuvel P, Mandl W, Kahn S, Hulshoff E. Functionally linked resting-state networks reflect the underlying structural connectivity architecture of the human brain. Human Brain Mapping, 2009;30(10), 3127–3141. https://doi.org/10.1002/hbm.20737
- Allman M, Tetreault A, Hakeem Y, Manaye F, Semendeferi K, Erwin M, Park S, Goubert V, Hof R. The von Economo neurons in frontoinsular and anterior cingulate cortex in great apes and humans. Brain Structure & Function, 2010;214(5–6), 495–517. https://doi.org/10.1007/s00429-010-0254-0
- Supekar K, Menon V. Developmental Maturation of Dynamic Causal Control Signals in Higher-Order Cognition: A Neurocognitive Network Model. PLoS Comput Biol, 2012;8(2), 1002374. https://doi.org/10.1371/journal.pcbi.1002374
- Vinod M, Uddin Q. (2010). Saliency, switching, attention and control: a network model of insula function. In Brain structure & function, 2010;214(5-6), 655-667. Springer. https://doi.org/10.1007/s00429-010-0262-0
- Darby R, Joutsa J, Burke J, Fox D. Lesion network localization of free will. 2019. https://doi.org/10.1073/pnas.1814117115
- Cohen A, Kaplan F, Moser J, Jenkins A, Wilkinson H. Impairments of attention after cingulotomy. Neurology, 1999;53(4), 819–824. https://doi.org/10.1212/wnl.53.4.819
- Cohen A, Kaplan F, Zuffante P, Moser J, Jenkins A, Salloway S, Wilkinson H. Alteration of intention and self-initiated action associated with bilateral anterior cingulotomy. Journal of Neuropsychiatry and Clinical Neurosciences, 1999;11(4), 444–453. https://doi.org/10.1176/jnp.11.4.444
- Wilkinson A, Davidson M, Davidson I. Bilateral anterior cingulotomy for chronic noncancer pain. Neurosurgery, 1999;45(5), 1129–1136. https://doi.org/10.1097/00006123-199911000-00023
- Zhou J, Greicius D, Gennatas D, Growdon E, Jang Y, Rabinovici D, Kramer H, Weiner M, Miller L, Seeley W. Divergent network connectivity changes in behavioural variant frontotemporal dementia and Alzheimer’s disease. Brain, 2010;133(5), 1352–1367. https://doi.org/10.1093/brain/awq075
- White P, Joseph V, Francis T, Liddle F. Aberrant salience network (bilateral insula and anterior cingulate cortex) connectivity during information processing in schizophrenia. Schizophrenia Research, 2010;123(2–3), 105–115. https://doi.org/10.1016/j.schres.2010.07.020
- Palaniyappan L, Mallikarjun P, Joseph V, White P, Liddle F. Reality distortion is related to the structure of the salience network in schizophrenia. Psychological Medicine, 2011;41(8), 1701–1708. https://doi.org/10.1017/S0033291710002205
- Whitfield-Gabrieli S, Thermenos W, Milanovic S, Tsuang T, Faraone V, McCarley W, Shenton E, Green I, Nieto-Castanon A, LaViolette P, Wojcik J, Gabrieli E, Seidman J. Hyperactivity and hyperconnectivity of the default network in schizophrenia and in first-degree relatives of persons with schizophrenia. Proceedings of the National Academy of Sciences of the United States of America, 2009;106(4), 1279–1284. https://doi.org/10.1073/pnas.0809141106
- Shao J, Meng C, Tahmasian M, Brandl F, Yang Q, Luo G, Luo C, Yao D, Gao L, Riedl V, Wohlschläger A, Sorg C. Common and distinct changes of default mode and salience network in schizophrenia and major depression. Brain Imaging and Behavior, 2018;12(6), 1708–1719. https://doi.org/10.1007/s11682-018-9838-8
- Castellanos X, Margulies S, Kelly C, Uddin Q, Ghaffari M, Kirsch A, Shaw D, Shehzad Z, Di Martino A, Biswal B, Sonuga-Barke S, Rotrosen J, Adler A, Milham P. Cingulate-Precuneus Interactions: A New Locus of Dysfunction in Adult Attention-Deficit/Hyperactivity Disorder. Biological Psychiatry, 2008;63(3), 332–337. https://doi.org/10.1016/j.biopsych.2007.06.025
- Sato R, Hoexter Q, Castellanos F, Rohde A. Abnormal Brain Connectivity Patterns in Adults with ADHD: A Coherence Study. PLoS ONE, 2012;7(9). https://doi.org/10.1371/journal.pone.0045671
- Posner J, Song I, Lee S, Rodriguez I, Moore H, Marsh R, Simpson B, Library O. Increased Functional Connectivity between the Default Mode and Salience Networks in Unmedicated Adults with Obsessive-Compulsive Disorder, 2016. https://doi.org/10.1002/hbm.23408
- Geng H, Li X, Chen J, Li X, Gu R. Decreased Intra- and Inter-Salience Network Functional Connectivity is Related to Trait Anxiety in Adolescents. Frontiers in Behavioral Neuroscience, 2016;9, 350. https://doi.org/10.3389/fnbeh.2015.00350
- Ordaz J, LeMoult J, Colich L, Prasad G, Pollak M, Popolizio M, Price A, Greicius M, Gotlib H. Ruminative brooding is associated with salience network coherence in early pubertal youth. Social Cognitive and Affective Neuroscience, 2017;12(2), 298–310. https://doi.org/10.1093/scan/nsw133
- Kaiser H, Andrews-Hanna R, Wager D, Pizzagalli A. Large-Scale Network Dysfunction in Major Depressive Disorder. JAMA Psychiatry, 2015;72(6), 603. https://doi.org/10.1001/jamapsychiatry.2015.0071