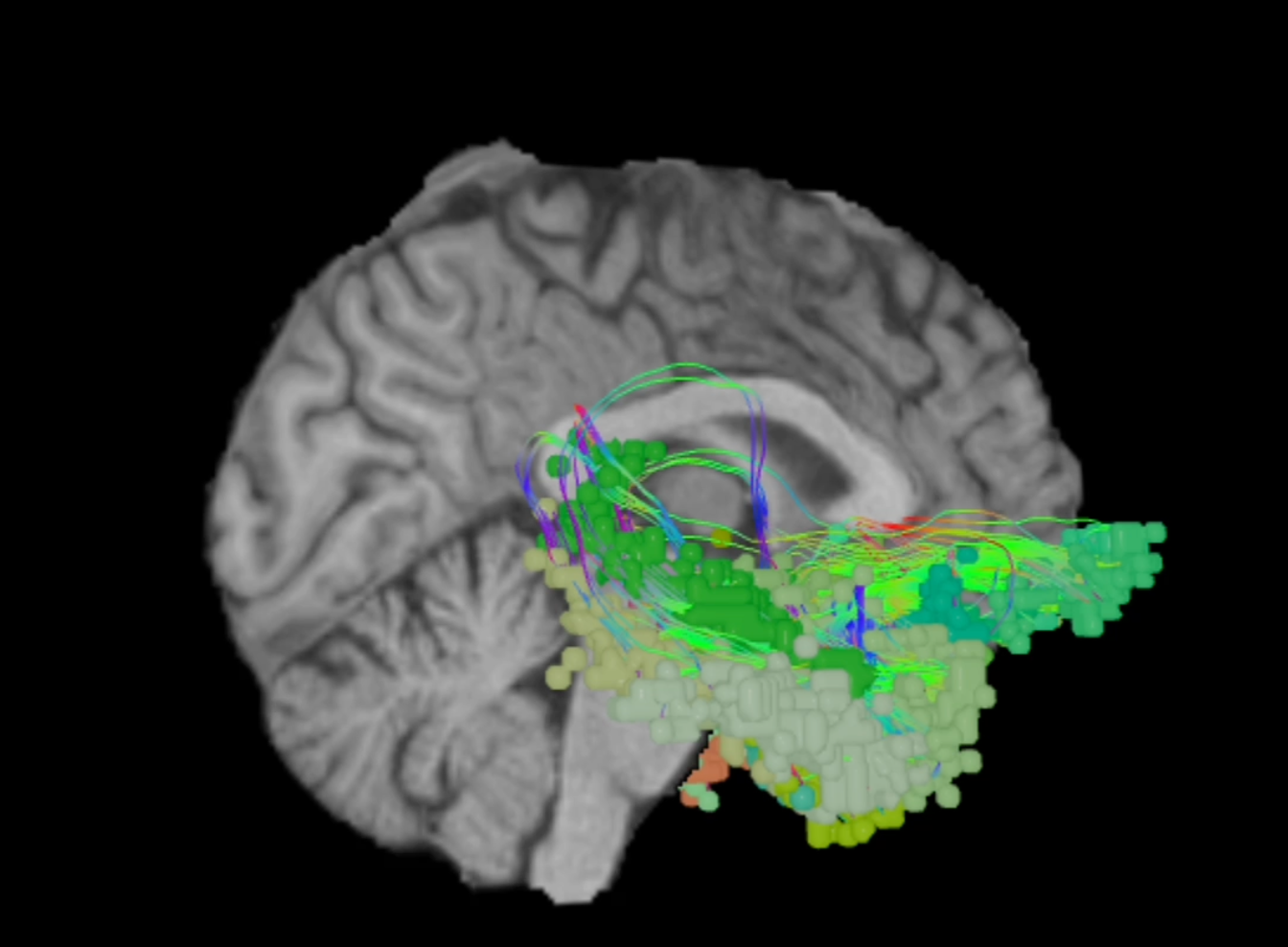
The brain has an extraordinary capacity to re-organize itself if specific areas are damaged. These areas, termed the rich-club, are highly efficient hubs that integrate information from multiple other regions5,6. Additionally, Ahsan and colleagues (2020) reported that PageRank centrality, a measure of connectedness to other regions, was a better predictor than other measures at identifying brain areas relevant to neurosurgery2. Considering that several of these classically identified areas are centrally located in network topology, damage to seemingly ineloquent regions alters the inputs to these hubs in ways that may or may not translate to functional impairments1.
Connectomics also provides a method to assess damage caused to major brain networks. Work performed by Hart and colleagues (2016) characterized consistent alterations in connectivity between a set of hub regions in pre-op temporal-parietal-occipital glioblastoma7. Functional connectivity changes have also been noted in patients suffering from TBI, stroke, and even hypertension8-10.
Variation between individuals is apparent whether one operates based on eloquence or centrality; otherwise, there would be no need for cortical mapping of awake patients during surgery1. Networks, as one may expect, differ just as much as individual regions. A study of a large pool of diffusion neuroimaging data from healthy subjects by Yeung and colleagues revealed a subset of individuals with strong hub activity in unexpected regions11. Glioma patients are much more heterogeneous and display distinct connectivity profile differences that could help inform resection that minimizes further harm11-13.
One such technique in development, supra-maximal resection, relies on the identification of essential hub areas to maximize post-surgical cortical organization while resecting more tissue than what is usually resected using traditional approaches6. In addition, brain networks can be used to apply techniques such as deep brain stimulation (DBS) more precisely. Evidence suggests that DBS can cause widespread effects that propagate along white matter. Using a combination of fiber tractography, fMRI, and advanced diffusion imaging modalities, it may be possible to apply DBS in more nuanced ways that target the network alterations of an individual patient13.
By mapping an individual patient's unique connectivity patterns, it may even be possible to predict functional deficits before surgery. In 2021, a team of researchers used Omniscient’s Quicktome software to see if the regions of strong hubness it identified could be correlated with neurological outcomes in 15 patients undergoing keyhole surgery12. Quicktome accurately predicted the functional deficits that emerged in seven of the eight patients with new post-operative impairments. The only outlier in this small study suffered a stroke shortly after surgery, potentially confounding results.
Quicktome was designed using machine-learning clustering techniques to rapidly analyze millions of data points from patient MRI scans based on connectivity profiles. This data is processed to generate an individualized map of a patient's brain networks. Quicktome has the added benefit of being cloud-based, so there is no need for new equipment as it seamlessly integrates into existing systems. Moreover, due to its cloud-based approach, surgeons are able to experience a hands-on, free trial of Quicktome by booking in a demo with their local representative.
Contact us now to get started or dig deeper into connectomics with our Connectome Academy.